Peritoneal mesothelioma is a rare type of cancer in the peritoneum, the lining of the abdomen. The primary cause of this cancer is ingesting asbestos fibers. Life expectancy for patients with peritoneal mesothelioma is two to six years. Unfortunately, because symptoms aren’t always clear, diagnosis often comes after cancer has spread.
What is peritoneal mesothelioma?
Peritoneal mesothelioma, which is the second most common type of mesothelioma, affects the lining of the abdomen.
Just like pleural mesothelioma, peritoneal mesothelioma is caused by exposure to asbestos. When someone ingests microscopic asbestos fibers, the tiny sharp particles get embedded in the lining of the abdomen (peritoneum). Over a course of 20 to 50 years or more, the fibers in the peritoneum cause mutations in the surrounding healthy mesothelial cells. Constant genetic damage makes these cells cancerous, forming tumors on the peritoneum.

- There are around 300-500 new peritoneal mesothelioma cases each year.
- Peritoneal mesothelioma is much less common than pleural mesothelioma, which has about 2,500 new diagnoses each year.
- Unlike pleural mesothelioma, peritoneal mesothelioma is more common in women than in men.
- Symptoms of peritoneal mesothelioma include unexplained weight loss, fever, abdominal pain and swelling.
- These symptoms are often subtle, so the cancer is often not diagnosed until after it has spread.
- This cancer is often diagnosed via biopsy, but may often be made with cytology fluid or other ways.
- There are no specific guidelines for treating peritoneal mesothelioma, and treatment often follows the same course as pleural mesothelioma.
What does your diagnosis mean?
Once doctors obtain a diagnosis, they can determine a treatment plan based on the cell type and how advanced the disease is. Peritoneal mesothelioma doesn’t have a standard staging system, so doctors classify the disease as either localized or advanced. Those with localized mesothelioma can generally handle more aggressive treatments.
What treatments work?
Surgery often offers the best chance for long-term survival. The cytoreduction with HIPEC has become the standard of care for peritoneal mesothelioma patients. The procedure removes tumors from the abdomen and treats the affected area with heated chemotherapy.
What’s the prognosis for this disease?
Thanks in part to HIPEC, peritoneal mesothelioma patients have a higher survival rate than those diagnosed with either pleural or pericardial mesothelioma. The survival time of some patients treated with this procedure has reached beyond 5 years.
Symptoms of peritoneal mesothelioma
Peritoneal mesothelioma symptoms are often caused by the buildup of fluid in the abdominal cavity (ascites) and the thickening of the lining of the abdomen (peritoneal thickening). The more advanced a patient’s mesothelioma is, the more pressure ascites and peritoneal thickening put on the internal organs in the abdominal cavity.
Common symptoms associated with peritoneal mesothelioma include:
|
|
The prognosis of peritoneal mesothelioma
Patients diagnosed with peritoneal mesothelioma typically enjoy a more favorable prognosis than those with other forms of the disease. Multiple studies have shown that patients who have cytoreduction with HIPEC experience significantly longer survival times than those who do not. Many patients now survive up to 5 years and some even live beyond 7 years. According to a study on its clinical results, most medical centers typically report a median survival rate ranging from 30 to 90 months after a cytoreduction with HIPEC.
Factors that can affect prognosis
Ultimately, a peritoneal mesothelioma patient’s prognosis is always unique because it’s based on factors that are individually specific.
These factors include:
- Age
- Overall health
- Cancer stage
- Mesothelioma cell type
Epithelioid cell type is the most common type when it comes to peritoneal mesothelioma. These cells generally respond favorably to treatment and have a lower chance of recurrence. Life expectancy for patients with epithelioid tumors tends to be longer than for other cell types.
The sarcomatoid cell type, which tends to be the most aggressive, is the rarest and doesn’t respond to treatment as well. There are also several rare subtypes of epithelioid and sarcomatoid cell types.
Biphasic mesothelioma tumors have both epithelioid and sarcomatoid cell types. As a result, prognosis and treatment options vary depending on which cell type is more common.
If you’ve been diagnosed with peritoneal mesothelioma, there are steps you can take to beat your prognosis. Living a healthy lifestyle, including eating healthy foods and getting plenty of exercise, can improve your overall health. If you’re healthy, you have a stronger immune system that can help fight mesothelioma.
The most important step a patient can usually take, however, is getting treatment from an experienced mesothelioma specialist. A specialist can select the most effective course of treatment for your specific cancer stage and mesothelioma cell type.
Discover How You Can Beat The Odds
Get your free mesothelioma guide and learn about your treatment options.
Get My Free GuideHow peritoneal mesothelioma develops
The primary cause of peritoneal mesothelioma is the ingestion of asbestos fibers through the digestive tract and into the abdomen. When people ingest these fibers, sharp asbestos particles become lodged in the peritoneum. These fibers continue to cause damage over time to the healthy, surrounding tissues in the peritoneum. Eventually, these fibers can lead to scar tissue developing, and fluid building up in the abdominal cavity. And over time, these fibers cause mutations in the tissues of the abdomen, eventually leading to cancerous tumors. Exposure can occur decades before symptoms begin to develop.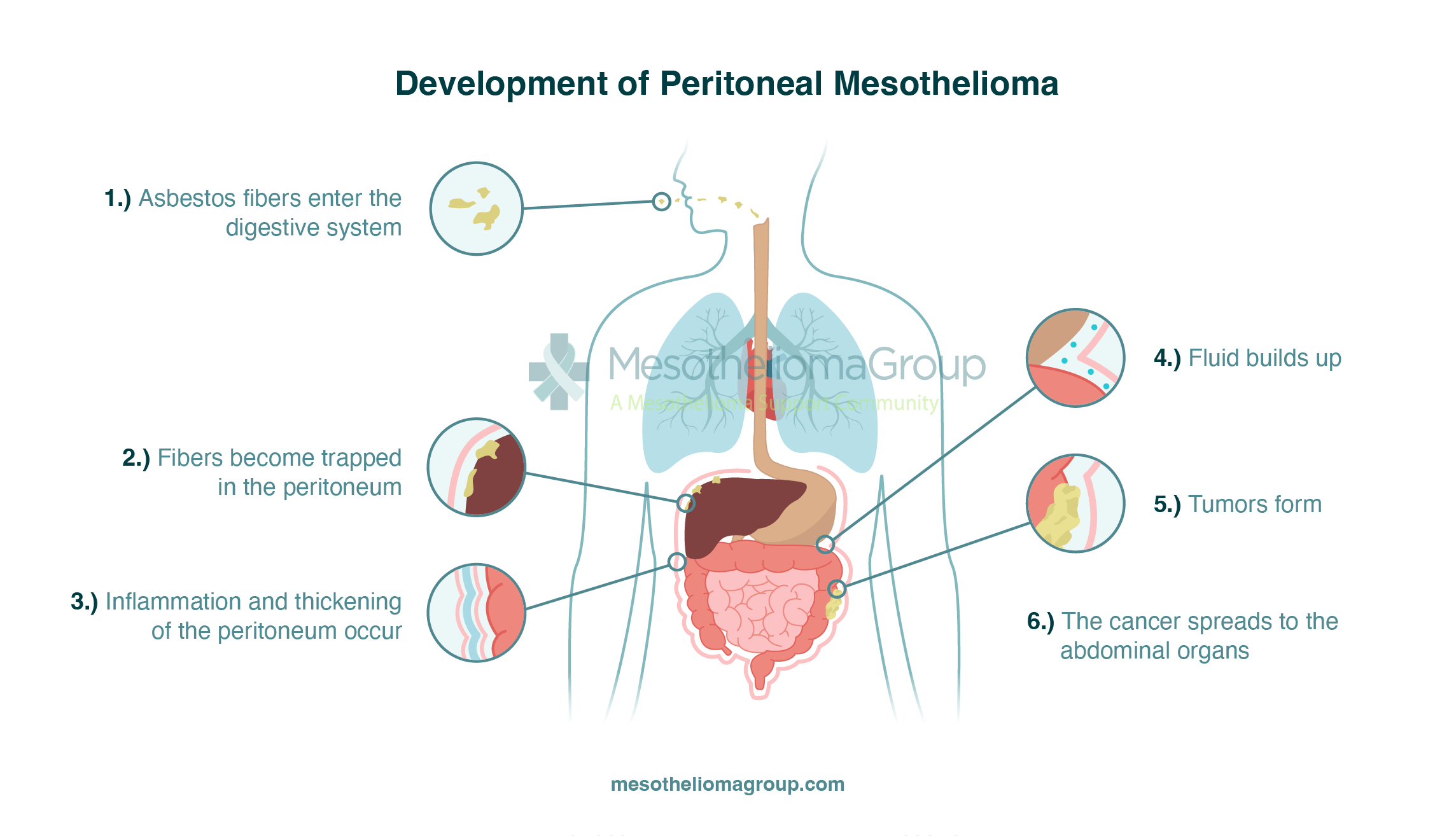
How peritoneal mesothelioma is diagnosed
The diagnosis of peritoneal mesothelioma typically begins with an x-ray. CT scans and MRIs aren’t used as often. The reason for this is that doctors can easily confuse mesothelioma on a CT scan or MRI with an abundance of gas and misdiagnose the disease as a result.
To confirm a peritoneal mesothelioma diagnosis, doctors will often order a diagnostic procedure called a peritoneoscopy. During this procedure, a surgeon inserts a small camera through a small incision in the patient’s abdomen. The camera helps the surgeon find and extract a tissue sample from the mesothelioma tumor in the abdomen. The surgeon sends the tissue sample to a lab where a pathologist tests the sample. Pathologists are doctors who specialize in the identification of different diseases. Sending the sample to a pathologist is necessary because peritoneal mesothelioma looks very similar to other cancers, even on a cellular level. Pathologists run several tests on the tumor sample to make sure that the suspected peritoneal mesothelioma diagnosis is correct.
Diagnostic steps for peritoneal mesothelioma
Physicians may rely on several tests to identify peritoneal mesothelioma. Patients may go through a progression similar to this:
- Symptoms develop as patients experience abdominal swelling and pain, unexplained weight loss, fever and other symptoms
- Physical evaluation from a physician to identify the next steps
- Imaging including X-rays, PET scans, CT scans and MRIs to check the peritoneal cavity
- Blood tests to check for specific cancer markers
- Biopsies to extract tumor tissue from the peritoneal cavity or surface
- Analysis of the biopsy and other tests to confirm a diagnosis and staging
How often is peritoneal mesothelioma misdiagnosed?
Unfortunately, peritoneal mesothelioma is so rare that it’s often misdiagnosed, especially since its clinical presentation can mimic other, more common conditions. Some conditions this cancer can be mistaken for include:
- Crohn’s disease
- Gallstones
- Hernia
- Inflammatory bowel disease
- Irritable bowel syndrome
- Ovarian cancer
It may be worthwhile to make an appointment with a specialist if you have been exposed to asbestos at any point in your life (especially since pleural mesothelioma can also be mistaken for other conditions, like lung cancer, influenza or COPD). Also, don’t be shy about getting a second opinion. Diagnosing this cancer at an early stage, before it has spread to surrounding lymph nodes, means a better chance at effective treatment.
Staging peritoneal mesothelioma
Staging cancer is an important step as it gives oncologists more information about what treatment options might work best based on how far the cancer has spread. For example, cancers in earlier stages have more options available, like surgery and radiation, than in later stages.
For peritoneal mesothelioma, many oncologists use the peritoneal cancer index (PCI). PCI is scored by dividing the abdomen into sections. Oncologists check for cancer and assign a number (0-3) if there is cancer in each section, then add those numbers to identify the stage. Like other systems, stage 1 indicates less advanced cancer, while stage 4 indicates more advanced cancer. 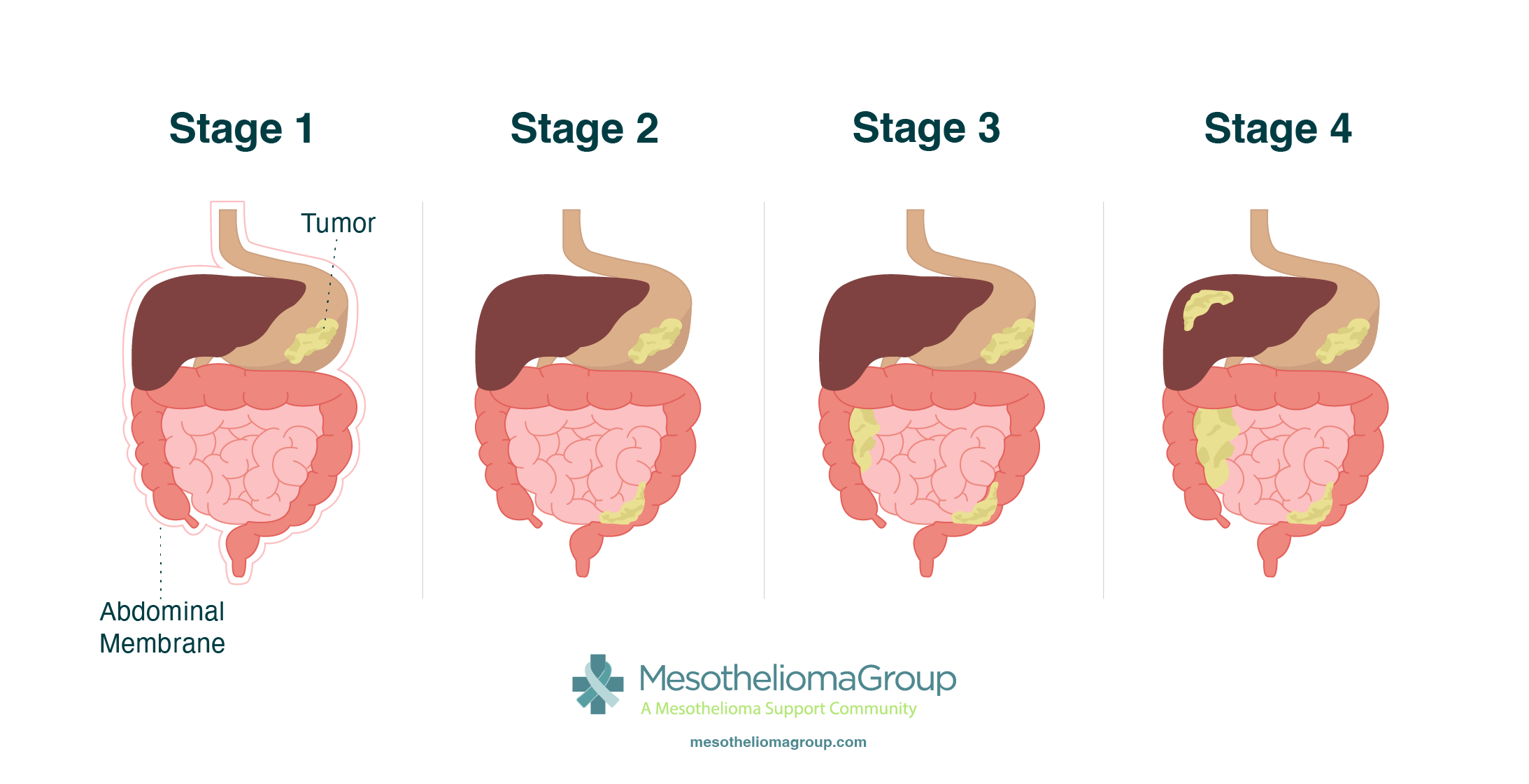
PCI score ranges for each stage are:
- Stage 1: 1-10
- Stage 2: 11-20
- Stage 3: 22-30
- Stage 4: 31-39
More generally, most oncologists rely on the TNM (tumor, nodes, metastasis) system to stage mesothelioma. For peritoneal mesothelioma, this system breaks mesothelioma into three stages rather than the four-stage system used for other cancers, including pleural mesothelioma.
Understanding the tumor node metastasis (TNM) system for staging mesothelioma
The TNM system, which is the most common system used by health systems for reporting, evaluates the growth of the primary tumor and how much it has spread to lymph nodes and other organs. This system was adapted to a three-stage approach for peritoneal mesothelioma to better stratify how far the disease has progressed compared to the traditional system.
When used for peritoneal mesothelioma, the TNM system measures:
- T: The disease burden in the peritoneum, using the PCI score
- N: Nodal metastasis within the abdomen
- M: Metastasis outside the abdomen
The 3 stages of peritoneal mesothelioma
Stage 1
At this stage, tumors are contained within a few sections of the abdomen. Cancer has not spread to nearby lymph nodes or distant areas of the body. Patients have more treatment options available at this stage, including a multi-modal surgical/chemotherapy approach.
Stage 2
In stage 2, tumors might be impacting more sections of the abdomen but have not yet spread to nearby lymph nodes or distant parts of the body.
Stage 3
By stage 3, tumors have spread throughout the abdomen. Cancer may have also spread to lymph nodes and distant parts of the body, including the lining of the small intestine and colon or the pleura (the lining of the lungs).
Treatments options for peritoneal mesothelioma
Patients diagnosed with peritoneal mesothelioma most often receive the above-mentioned cytoreductive surgery with hyperthermic intraperitoneal chemotherapy (HIPEC). This combination of treatments is also generally known as a multimodal therapy. Dr. Paul Sugarbaker, one of the most respected mesothelioma specialists in the U.S., pioneered the addition of HIPEC to the procedure.
The addition of HIPEC to cytoreductive surgery has led to significant leaps in life expectancy for peritoneal mesothelioma patients.
Cytoreductive surgery
Cytoreductive surgery is a procedure surgeons use to remove mesothelioma tumors that are in and around the abdominal cavity. A surgeon removes any visible signs of cancer. The surgeon may also remove nonessential organs affected by mesothelioma. The procedure itself is highly complex and can take up to 10 hours to complete.
Heated intraperitoneal chemotherapy (HIPEC)
HIPEC is a heated mixture of chemotherapy drugs introduced into the abdominal cavity shortly after the cytoreductive surgery is completed. The goal of HIPEC is to kill any microscopic traces of mesothelioma that may remain after surgery.
The mixture of drugs is typically heated to a temperature of 104 – 107 degrees and circulated inside the abdominal cavity for a maximum of 2 hours. This allows the chemotherapy enough time to be absorbed into the microscopic cancer cells and increases the cancer-killing effectiveness of the drugs.
HIPEC may be given once, shortly after the surgical procedure, or as many times as the surgeon sees fit in the weeks following the surgery. One recent study reported that patients who receive repeated HIPEC after a single cytoreductive surgery experienced a longer survival time (approximately 80 months) than those who only received it once (27.2 months).
Paracentesis
A paracentesis involves the use of a needle or catheter to remove the buildup of fluid in the abdominal cavity. This procedure is mainly used to relieve any discomfort or pain caused by symptoms of peritoneal mesothelioma.
Palliative care for peritoneal mesothelioma
Palliative care is not a curative treatment to eliminate cancer. Rather, the goal is to improve a patient’s quality of life by managing their pain and relieving symptoms.
For patients with peritoneal mesothelioma, palliative care options may include:
- Intervention to remove excess fluids
- Interventions to reduce weight loss
- Medication management
- Radiation therapy to target painful tumors
- Surgery to remove obstructive tumors
Our Patient Help Team has access to a network of mesothelioma specialists and can help connect you to an experienced professional now.
Peritoneal mesothelioma FAQs
What causes peritoneal mesothelioma?
The primary cause of peritoneal mesothelioma is asbestos exposure.
How quickly does peritoneal mesothelioma spread?
Peritoneal mesothelioma can take decades to appear following asbestos exposure. Certain cell types, like sarcomatoid and biphasic mesotheliomas, are very aggressive and metastasize to other areas of the body quickly. Other cell types, like epithelioid mesothelioma, are less aggressive. Early diagnosis from an oncologist means more opportunities for the best treatment options.
What is the survival rate for peritoneal mesothelioma?
Around 25% of patients with peritoneal cancer survive three years beyond diagnosis. Life expectancy for patients with peritoneal mesothelioma is two to six years. Early diagnosis and mesothelioma treatment can increase life expectancy.
Is peritoneal mesothelioma painful?
Peritoneal mesothelioma often does not have noticeable symptoms until after it has spread throughout the abdominal cavity. After it has spread, abdominal pain is a common symptom.
Can you go into remission with peritoneal mesothelioma?
Though rare, remission is possible if peritoneal mesothelioma is caught and treated early. Treatments like systemic chemotherapy, which delivers cancer-fighting drugs throughout the body, radiation and surgery can help.
What resources are available to help my loved one with mesothelioma?
To learn more about mesothelioma or to find information on cancer treatment, clinical trials or new treatment options, contact Mesothelioma Group at (800) 333-8975.
- Sources
-
Repeat Cytoreductive Surgery and Heated Intraperitoneal Chemotherapy May Offer Survival Benefit for Intraperitoneal Mesothelioma: A Single Institution Experience. Retrieved on October 20, 2014 from http://link.springer.com/article/10.1245/s10434-013-3341-7#page-1.
-
Clinical Results of Cytoreduction and HIPEC for Malignant Peritoneal Mesothelioma. Retrieved on October 17, 2014 from http://link.springer.com/chapter/10.1007/978-0-387-48993-3_22.
-
Clinical Results of Cytoreduction and HIPEC for Malignant Peritoneal Mesothelioma. Retrieved on October 20, 2014 from http://jco.ascopubs.org/content/27/36/6237.short.
-
Peritoneal Mesothelioma Treatment. Retrieved on October 17, 2014 from http://www.cancerresearchuk.org/about-cancer/type/mesothelioma/treatment/types/peritoneal-mesothelioma-treatment.
-
Diseases and Conditions: Mesothelioma. Retrieved on October 17, 2014 from http://www.mayoclinic.org/diseases-conditions/mesothelioma/basics/definition/con-20026157.
-



